Episode Details
Back to Episodes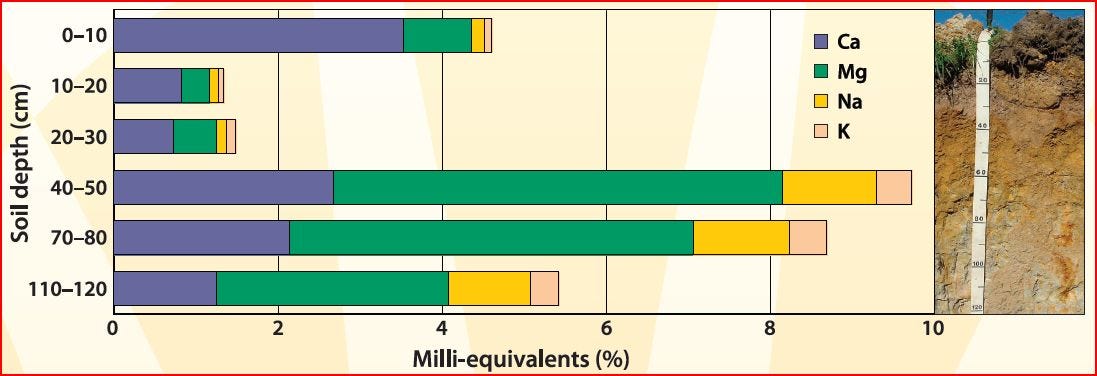
Cation Exchange Capacity for Gardeners
Description
Debbie Flower, America’s Favorite Retired College Horticulture Professor, brought up the subject of “Cation Exchange Capacity” back in Episode 193 of the Garden Basics with Farmer Fred podcast, while discussing the limited lifespan of bagged worm castings. That alone should pique your interest. Go ahead and listen to that segment or read the transcript. I’ll wait.
In the podcast in this newsletter, Debbie goes into depth about cation exchange capacity. If you were a high school chemistry class wizard, you’re excited to learn how the electronic charges in the soil aid and abet the feeding of plants. The rest of us need a bit more help (I took high school chemistry a couple of times. I still don’t get it.).
Here’s an almost easy to understand explanation of the chemistry going on beneath our feet. From Australia’s Katharine Brown (The University of Western Australia) and Jeremy Lemon (Department of Agriculture and Food, Western Australia), entitled, “Cations and Cation Exchange Capacity”. At the very least, you’ll learn about “meq”. Again, this is why this newsletter is entitled the way it is.
Cations and Cation Exchange Capacity
Key Points
* Cation exchange capacity (CEC) is the total capacity of a soil to hold exchangeable cations.
* CEC is an inherent soil characteristic and is difficult to alter significantly.
* It influences the soil’s ability to hold onto essential nutrients and provides a buffer against soil acidification.
* Soils with a higher clay fraction tend to have a higher CEC.
* Organic matter has a very high CEC.
* Sandy soils rely heavily on the high CEC of organic matter for the retention of nutrients in the topsoil.
*
Background
Cation exchange capacity (CEC) is a measure of the soil’s ability to hold positively charged ions. It is a very important soil property influencing soil structure stability, nutrient availability, soil pH and the soil’s reaction to fertilisers and other ameliorants (Hazleton and Murphy 2007).
What are exchangeable cations?
The clay mineral and organic matter components of soil have negatively charged sites on their surfaces which adsorb and hold positively charged ions (cations) by electrostatic force. This electrical charge is critical to the supply of nutrients to plants because many nutrients exist as cations (e.g. magnesium, potassium and calcium). In general terms, soils with large quantities of negative charge are more fertile because they retain more cations (McKenzie et al. 2004) however, productive crops and pastures can be grown on low CEC soils.The main ions associated with CEC in soils are the exchangeable cations calcium (Ca2+), magnesium (Mg2+), sodium (Na+) and potassium (K+) (Rayment and Higginson 1992), and are generally referred to as the base cations. In most cases, summing the analysed base cations gives an adequate measure of CEC (‘CEC by bases’). However, as soils become more acidic these cations are replaced by H+, Al3+ and Mn2+, and common methods will produce CEC values much higher than what occurs in the field (McKenzie et al. 2004). This ‘exchange acidity’ needs to be included when summing the base cations and this measurement is referred to as effective CEC (ECEC).
Measuring CEC
Different laboratories use various methods to measure CEC, and can return contrasting results depending on the fraction of the soil measured. In Australia, some laboratories measure CEC directly and others calculate it as CEC by bases. Cation